Short Note: Repurposing Drugs—Using a Mental Health Drug for the Treatment of Alzheimer’s Disease
Repurposing drugs for serious illnesses will improve lives in this time of lack of research funding
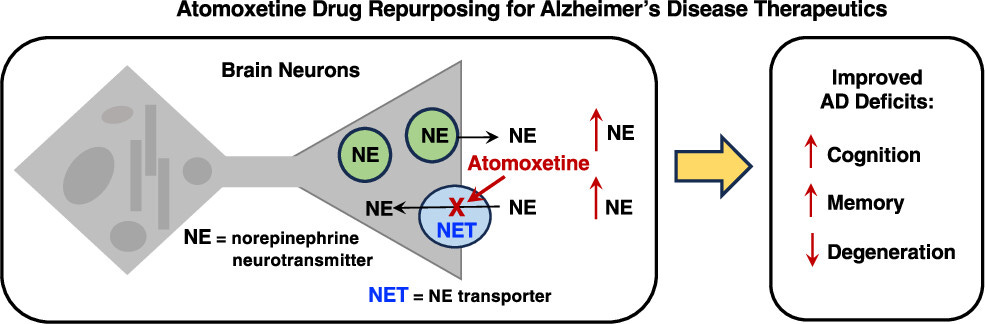
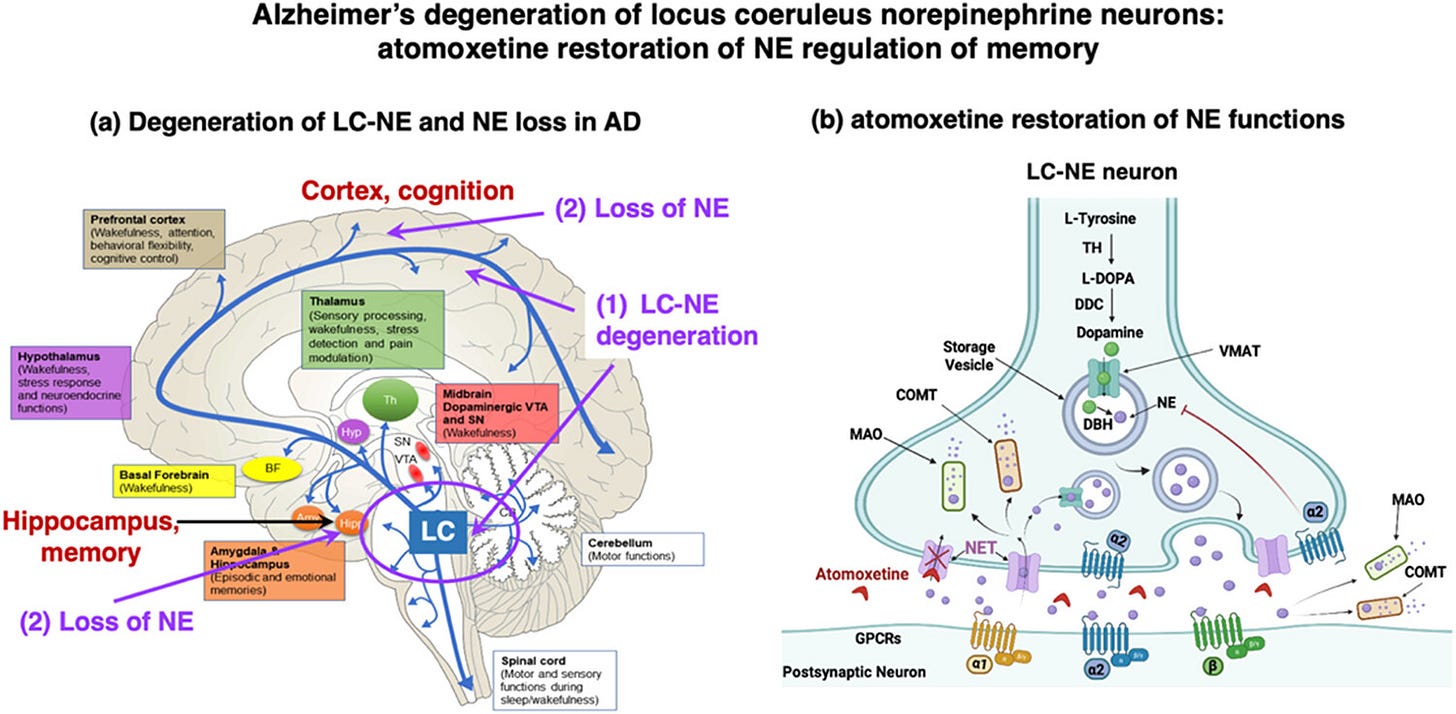
Atomoxetine is a drug specifically used to treat Attention Deficit Hyperactivity Disorder, and it is being investigated for the treatment of Alzheimer’s Disease. While the drug is also used to treat depression, it has recently gained notice in the scientific community as a potential Alzheimer’s drug. In an article published late last year in the American Chemical Society journal, ACS Pharmacology and Translational Science, by 16 researchers from three US universities, we learn that the drug atomoxetine improved cognition and slowed the course of dementia in mild to moderately impaired Alzheimer’s patients. This bears mentioning because of the lack of coverage in everyday science news.
While the research is still early, the finding is important since government funding for new research is drying up. It is considered a ‘pipedream’ by the majority of US researchers to get funding from the Federal government for any research. Shedding light on this research might drive big pharma to pick up the slack. However, the funding cuts by DOGE and the regime will make the training of new researchers in US universities more difficult.
The drug atomoxetine is well-tolerated in clinical trials. While only in Phase II trials, atomoxetine has also been shown to be effective in the treatment of Parkinson’s disease. Because of the similarities between Alzheimer’s disease and Parkinson’s disease, it is a safe bet to say that the final form of this new drug will most likely treat some forms of Alzheimer’s disease. Moreover, the researchers concluded: (1) synaptic functions improved in Alzheimer’s patients, (2) animal studies further corroborate these findings, (3) the safety in the doses administered leaves many in the medical community hopeful, as well.
Whether we see atomoxetine or its newer derivatives as a treatment option in the near-to-mid-future will depend on how the FDA is restructured by the regime. None of what seems to be going on right now gives many of us much hope. This era will prove to be one of the most difficult in the collective memories for all of us.
—-
Demsey, Laura L., et al. “Atomoxetine Drug Properties for Repurposing as a Candidate Alzheimer’s Disease Therapeutic Agent.” ACS Pharmacology & Translational Science 8.11 (2025): 3757-3772.
Fedder, Douglas, et al. “Atomoxetine.” StatPearls, StatPearls Publishing, 27 March 2023.